A comprehensive, evidence‑based Clinical Guideline for the Diagnosis and Treatment of Fibrolamellar Carcinoma (FLC) has been released online ahead of print publication in Hepatology, the flagship journal of the American Association for the Study of Liver Diseases. This landmark paper represents the first formal, multidisciplinary consensus guideline developed specifically for FLC, a rare liver cancer that primarily affects adolescents and young adults.
The guideline was authored by a national panel of 20 physicians from 15 leading academic cancer centers across the United States, spanning pediatric and adult oncology, hepatology, surgery, pathology, and interventional radiology. The effort was co‑led by Drs. Allison O’Neill (Dana‑Farber Cancer Institute and Boston Children’s Hospital) and John Gordan (University of California, San Francisco), with financial support from the Fibrolamellar Cancer Foundation (FCF). Several panelists also serve on FCF’s Medical and Scientific Advisory Board.
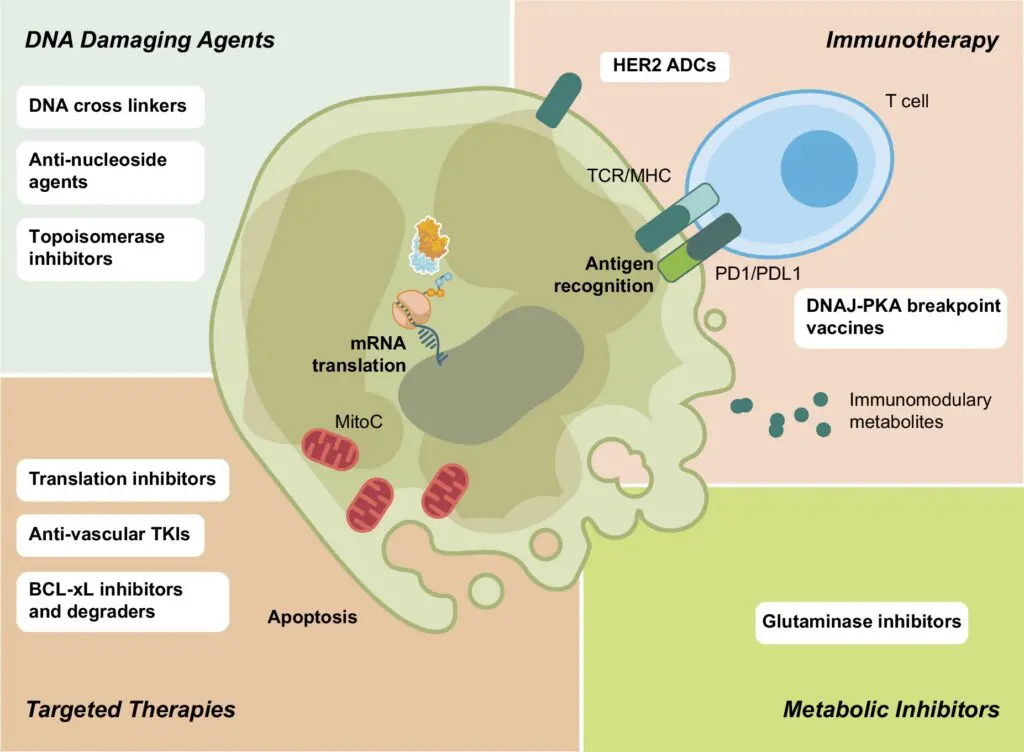
Central to the guideline is the firm recognition that FLC is biologically distinct from conventional hepatocellular carcinoma (HCC) and should not simply be treated using HCC‑based approaches. The authors provide consensus recommendations across five major domains: diagnosis, surgery, locoregional therapy, systemic therapy, and psychological, social, and palliative care. Key recommendations include routine molecular testing to confirm the nearly universal DNAJB1‑PRKACA gene fusion that defines FLC, aggressive and repeated surgical intervention when feasible, and the strategic use of locoregional therapies to enable or complement surgery.
Hepatology, March 2026
Acknowledging the absence of clearly curative systemic therapies for most patients, the guideline strongly emphasizes early referral to clinical trials—not as a last resort, but as a core component of standard care, including in the adjuvant setting after surgery. While outlining current best practices, the publication also highlights emerging areas of progress, including immunotherapy combinations, precision approaches such as antibody‑drug conjugates, and new biomarkers for disease monitoring.
Together, these guidelines provide physicians, patients, and caregivers with a clear snapshot of today’s best FLC care, while underscoring the urgent need for continued research to improve long‑term outcomes for this rare and devastating disease.
Frequently Asked Questions: New Clinical Guidelines for Fibrolamellar Carcinoma (FLC)
FLC is a rare cancer that behaves very differently from more common liver cancers. Historically, many patients were treated using approaches designed for hepatocellular carcinoma (HCC) or pediatric hepatoblastoma, which often are not effective for FLC. These guidelines formally recognize FLC as a distinct disease and establish FLC‑specific best practices to improve consistency, decision‑making, and outcomes.
Because fibrolamellar carcinoma (FLC) is extremely rare, creating reliable clinical guidelines required a careful, transparent, and consensus‑driven process that combined published evidence with deep expert experience.
Who created the guidelines?
The guidelines were developed by a multidisciplinary panel of 20 nationally and internationally recognized FLC experts. The group included medical oncologists, pediatric oncologists, liver and transplant surgeons, pathologists, radiation and interventional radiologists, and specialists in palliative care and clinical social work. This ensured that every aspect of FLC care—from diagnosis and treatment to supportive care—was represented.
What evidence was used?
The panel relied on three complementary sources of evidence:
- A comprehensive review of the published medical literature, including hundreds of peer‑reviewed articles, case reports, and clinical studies identified through major medical databases.
- Structured, one‑on‑one interviews with all 20 panelists to capture real‑world experience, including what has worked, what has failed, and how treatment decisions are made in practice.
- Additional targeted literature searches based on therapies and approaches raised during expert interviews, ensuring that all reported treatments showing benefit in FLC were considered.
How were recommendations decided?
Treatments were evaluated using a formal voting process to measure expert consensus. Recommendations were labeled “strong” or “weak” based on how many experts supported them, and therapies without sufficient agreement were excluded altogether. This ensured that every recommendation reflects meaningful consensus rather than individual opinion.
Why does this approach matter for patients?
Because large randomized trials are rare in FLC, expert experience plays a critical role. This rigorous methodology ensures that the guidelines are:
- Evidence‑informed
- Expert‑validated
- Consensus‑based
- Practical for real‑world care
- Transparent in how conclusions were reached.
This process makes the guideline the most authoritative and trustworthy clinical resource available today for patients, caregivers, and physicians facing FLC.
The recommendations span five major areas:
- Diagnosis
- Surgery
- Locoregional therapies (tumor‑directed treatments within the liver)
- Systemic therapies (chemotherapy, targeted therapy, immunotherapy)
- Psychological, social, and palliative care.
This reflects a whole‑person approach to FLC care, not just tumor treatment.
A major recommendation is routine molecular testing to confirm the presence of the DNAJB1::PRKACA gene fusion, which is found in nearly all FLC tumors and is considered a defining feature of the disease. The guidelines also encourage broader genomic testing when appropriate, as well as the use of emerging blood‑based biomarkers to help monitor disease activity.
Surgery remains the single most effective treatment for extending survival in FLC. The Guidelines recommend aggressive surgical approaches, including repeat resections, staged procedures, and debulking surgeries when appropriate. Unlike many other cancers, selected patients with metastatic or recurrent FLC may still benefit significantly from surgery.
Yes. The panel recommends that liver transplantation be considered for patients with unresectable disease confined to the liver, even if they do not meet traditional criteria used for other liver cancers. This reflects growing evidence that transplantation can meaningfully extend survival for some FLC patients.
Locoregional therapies are treatments that directly target tumors in the liver, typically administered by interventional radiologists. These include procedures such as Y‑90 radioembolization (TARE), chemoembolization (TACE), stereotactic body radiation therapy (SBRT), and tumor ablation techniques. They are often used to control disease, relieve symptoms, or shrink tumors to make surgery possible.
The guideline acknowledges that FLC generally responds poorly to many standard systemic therapies. However, it highlights recent progress and identifies several drug combinations that have shown promise in clinical practice, particularly for:
- Shrinking tumors before surgery
- Treating advanced or metastatic disease
- Supporting treatment after surgery.
These recommendations are based on emerging evidence and expert experience rather than large prospective trials.
Immunotherapy, particularly immune checkpoint inhibitors, has shown encouraging results in a small number of FLC patients. The guidelines highlight this as an area of active progress, including early clinical trials combining immune checkpoint inhibitors with vaccines targeting the FLC‑specific fusion protein. While not yet a cure, these approaches represent one of the most hopeful frontiers in FLC treatment.
Yes—very strongly. The panel recommends that clinical trials be offered early, not only as a last resort. This includes trials for patients who cannot undergo surgery and trials used after surgery to reduce the risk of recurrence. Research participation is essential to improving care for current and future FLC patients.
The guidelines describe today’s best available practices, but they also emphasize that there are still no universally curative therapies for most patients. Medicine is evolving, and recommendations will change as new evidence emerges. The document is meant to inform decision‑making and encourage expert consultation and research involvement.
Patients and families can use the guidelines to:
- Ask informed questions of their care team
- Seek evaluation at centers with FLC expertise
- Understand why certain tests or treatments are recommended
- Advocate for clinical trial participation when appropriate.
Yes. As new research emerges (especially from FLC‑focused clinical trials) the recommendations will evolve. FCF and the medical community view this guideline as a foundation, not as an endpoint.
More detailed information and specific expert recommendations from key sections of the guideline document are summarized in the linked pages below:
The full text of the guidelines can be read and downloaded here.
Note: This study was funded by a grant from FCF.
